
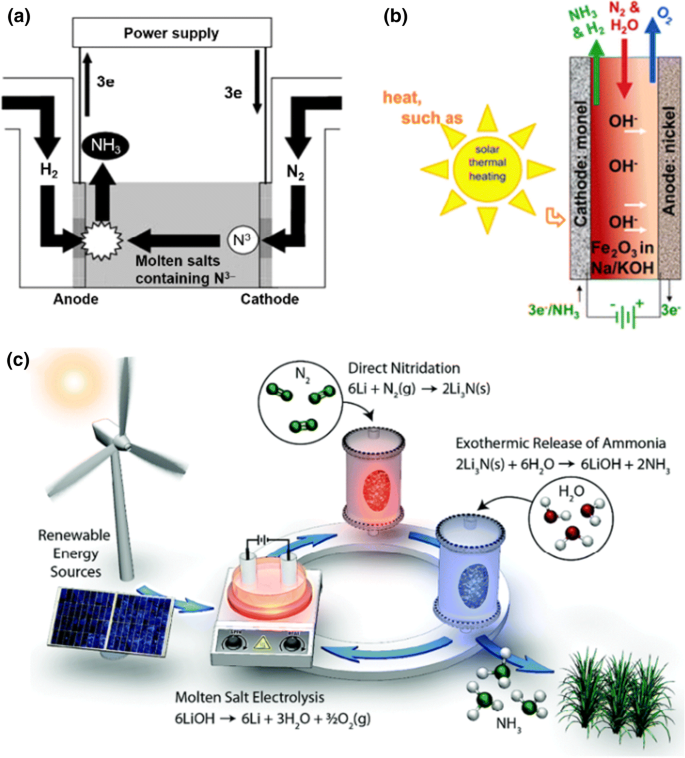
This will kill fishes in the rivers and other water streams. Ammonia dissolves in water and water become basic.Adding NH 3(g) gas into atmosphere will pollute air.To prevent coagulation of natural latex.Įnvironmental pollution of Ammonia IndustryĪmmonia industry may cause several environmental pollution due to its products and physical conditions.Then very low temperature can't be applied.Īmmonia has so many uses in industrial scale and domestic scale.It means ammonia product in a unit time (per day , Therefore, the reaction rate will fall.Decrease of energy causes decrease of number of molecules which have activation energy.To Happen a reaction, moleculers should haveĪctivaton energy.Decrease of heat results decrease of energy in reactants.Therefore removing heat from the reactor will bring equlibrium constant to the right giving large.Forward reaction of NH 3 production is exothermic reaction.Decreasing Temperature means of decrease of Heat in the reactor.And also adding more reactants will cost more money.Ĭan obtain more ammonia, decreasing temperature?.Therefore affordable devices have to be applied for high pressure. But more reactants will increase the pressure in the reactor.Excess reactants will bring equilibrium constant to the right.Equilibrium of hydrogen and nitrogen reaction Can obtain more ammonia, increasing reactants?
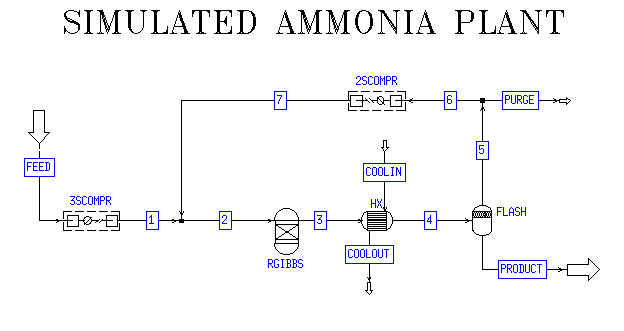
Unreacted nitrogen and ammonia is resent to the react. When product is being cooled ammonia becomes to liquidĪs the final product liquid ammonia is separated. Therefore Around 200 atm pressure should be maintained inside the reactor. Also to generate high pressure, cost is high. So increasing the pressure will bring the reaction more forward (producing more NH 3).īut Equipments which are affordable to high pressure are expensive. With that, volume is decreased due to decrease of Therefore pressure is decreased due to reduction of number of molecules. ( one N 2 molecule + three H 2 molecules). In forward reaction, two molecules are given by reacting four molecules. By considering both product amount and production rate, temperature is decided.Ĥ00 0C - 450 0C temperature is applied. So, manufactures have to wait much time to But having low temperature will reduce the Therefore using low temperature will increase the forward reaction and give higher percentage ofĪmmonia. K 2O (s), Mo or Ca are used as catalyst developers.įorward reaction is a exothermic one. K 2O (s), Al 2O 3(s) are used as catalyst developers.
Then air can be converted to liquid phase. Pressure and temperature of air is increased. Hydrogen is derived from natural gas or petroleum refining.Reactions involving in ammonia production is explained with each step.

In this section, we discuss how raw materials are converted to ammonia and what conditions (optimum conditions) should be maintained Manufacturing process of ammonia, Haber process Ammonia is basic gas and it is found as aqueous ammonium hydroxide solution in the laboratory. Ammonia is a very toxic compoundĪnd have to use it very carefully to prevent injuries.Īmmonia is a very toxic, sharp suffocating odor, colourless gas. Laboratories, ammonia is met as aqueous ammonium hydroxide (NH 4OH) solution.


 0 kommentar(er)
0 kommentar(er)
